
ACUITY Trial
Overview of the ACUITY Phase 2 trial
Phase 2 Results of Privosegtor for the treatment of optic neuritis
The Phase 2 ACUITY (Acute OptiC NeUrITis of DemYelinating Origin) trial was a randomized, double-blind, placebo-controlled, multi-center trial, designed to evaluate Privosegtor administered intravenously once-daily for five days in patients with an acute episode of optic neuritis also receiving IV corticosteroids. The study randomized 36 patients with recent onset of visual loss symptoms of unilateral acute optic neuritis of which 33 patients received treatment and were included in the pre-specified modified intent-to-treat (mITT) analysis.
In the ACUITY trial, Privosegtor achieved the primary safety endpoint and showed functional vision improvement and neuroprotective biological and anatomical benefits in patients suffering from optic neuritis. clinicaltrials.gov identifier: NCT04762017
Primary Endpoint was Safety-Based:
The percentage of patients with a shift from normal (baseline) to abnormal in electrocardiogram (ECG) parameters after study drug administration until Visit 4 (Day 15) was measured to evaluate cardiac safety. The results didn’t show an increase of patients with abnormal ECG parameters in Privosegtor versus placebo.
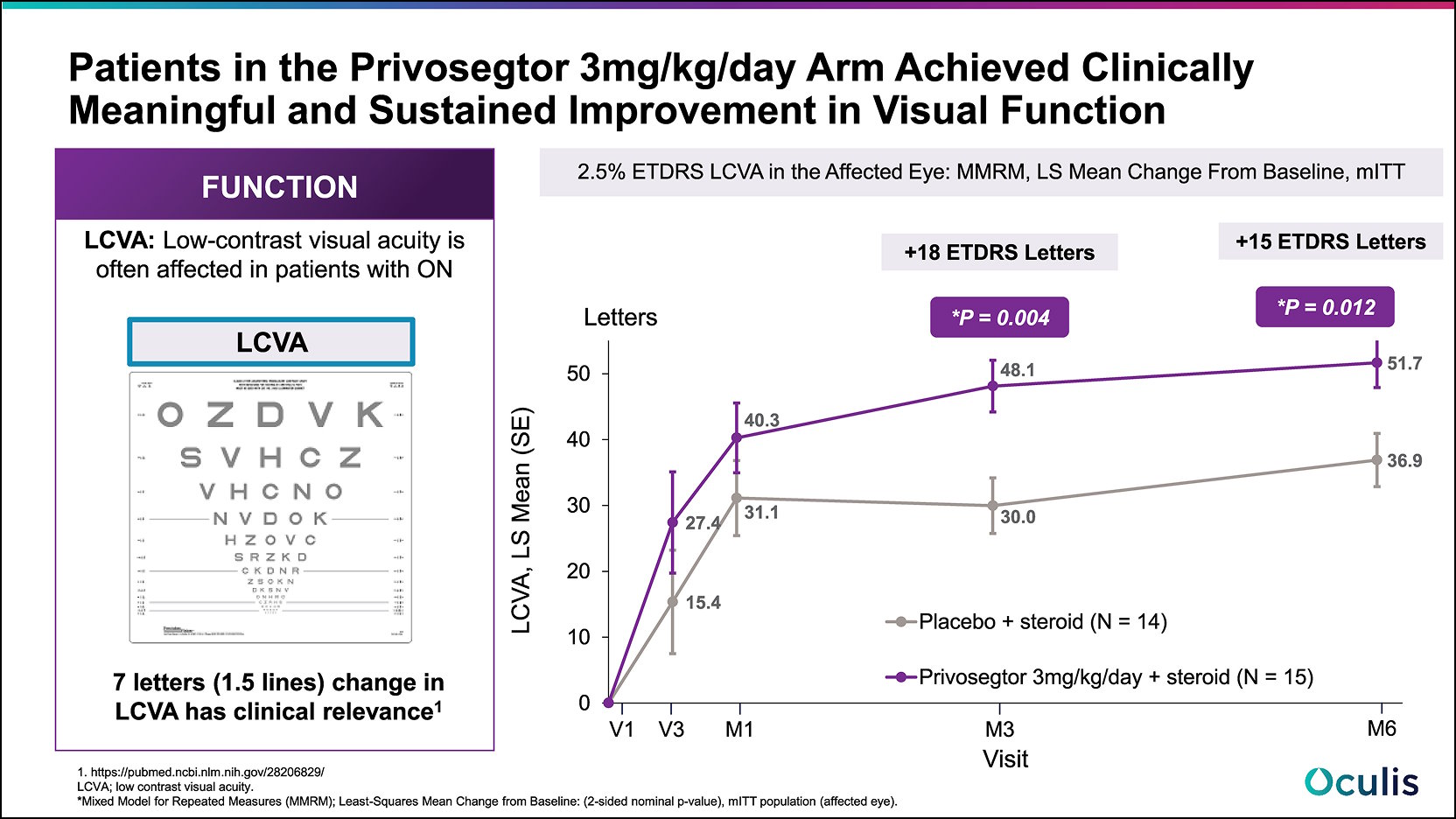
Secondary Efficacy Endpoint Assessed Changes in Visual Function:
Changes in 2.5% ETDRS low contrast letter acuity (LCVA) were measured to assess visual function improvement. Results showed:
- A favorable difference in LCVA mean change from baseline of approximately 18 letters at month 3 and approximately 15 letters at month 6 with Privosegtor + steroid compared to placebo + steroid, with p-values^ of 0.004 and 0.012 at 3 and 6 months, respectively.
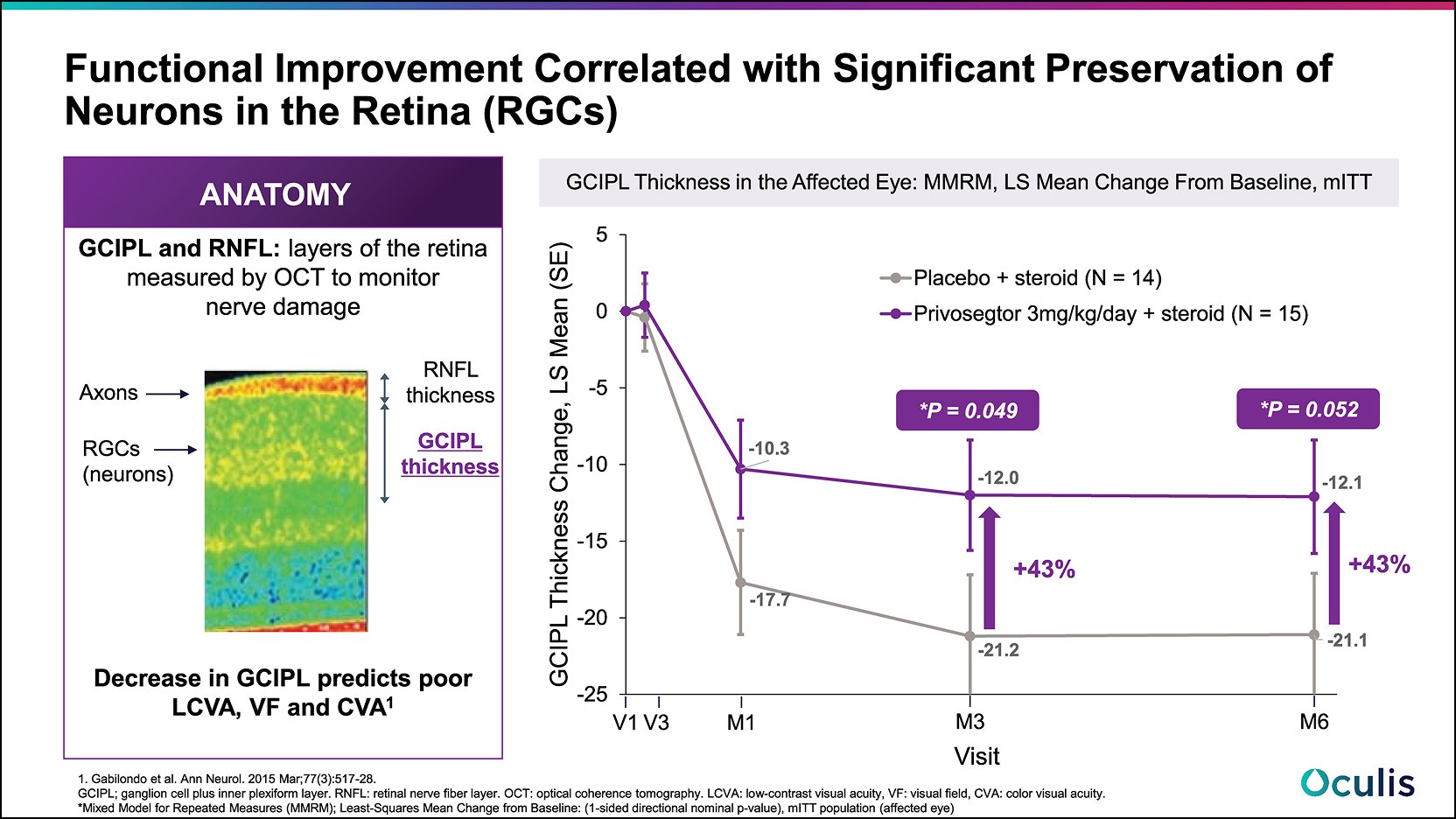
Secondary Efficacy Endpoints Assessed Changes in Retinal Structure:
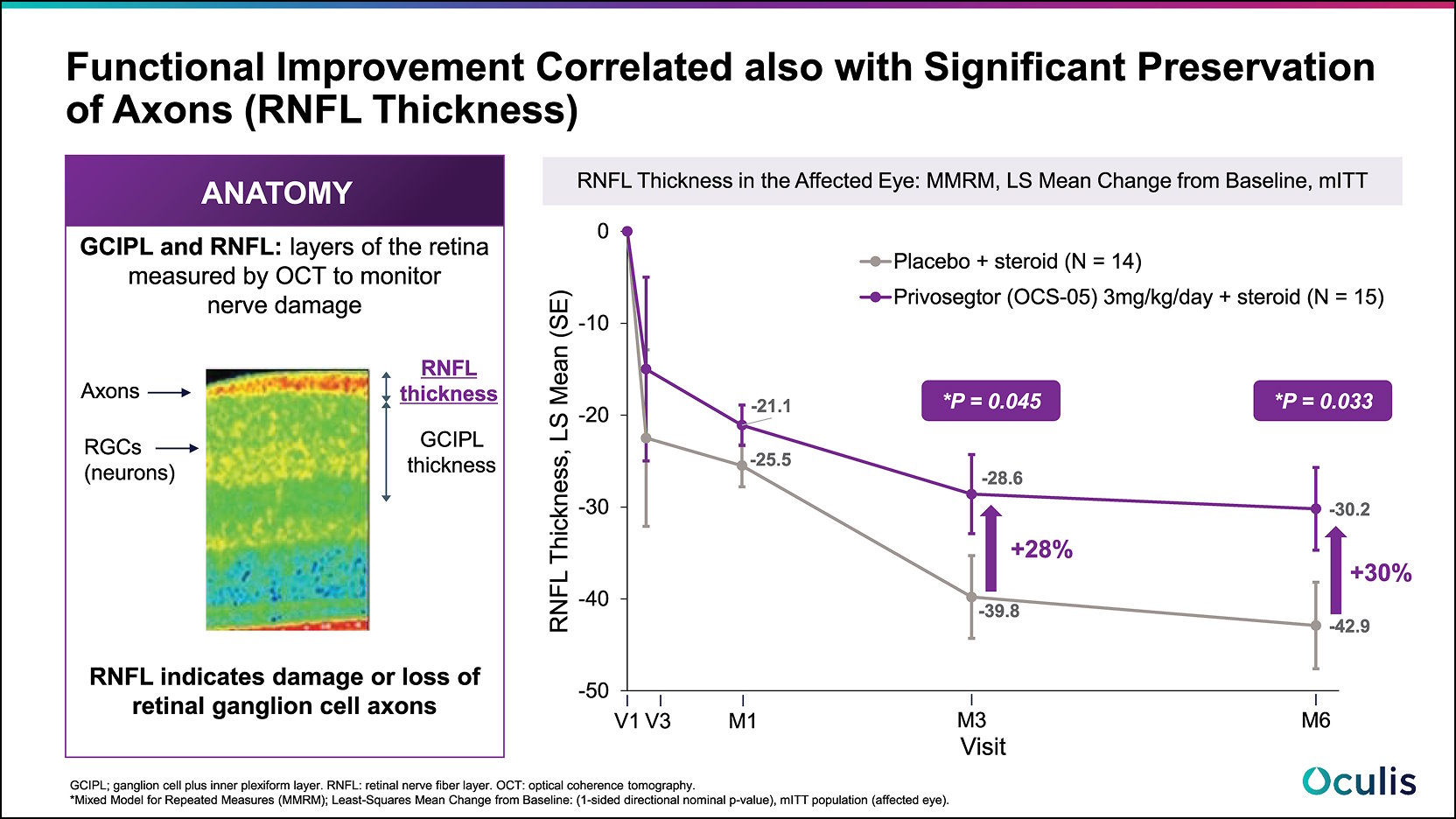
Optical Coherence Tomography (OCT) imaging was used to objectively measure the thickness of two different retinal segments in the affected eye to evaluate the potential neuroprotective effects of Privosegtor compared to placebo: 1) Ganglion Cell-Inner Plexiform Layer (GCIPL) and 2) Retinal Nerve Fiber Layer (RNFL). Results showed:
- A 43% improvement in GCIPL thickness mean change from baseline in favor of Privosegtor + steroid compared to placebo + steroid at month 3 which was maintained through month 6 with p-values* of 0.049 and 0.052 at 3 and 6 months, respectively.
- A 28% improvement in RNFL thickness mean change from baseline in favor of Privosegtor + steroid compared to placebo + steroid at month 3 reaching 30% improvement at month 6 with p-values* of 0.045 and 0.033 at 3 and 6 months, respectively.
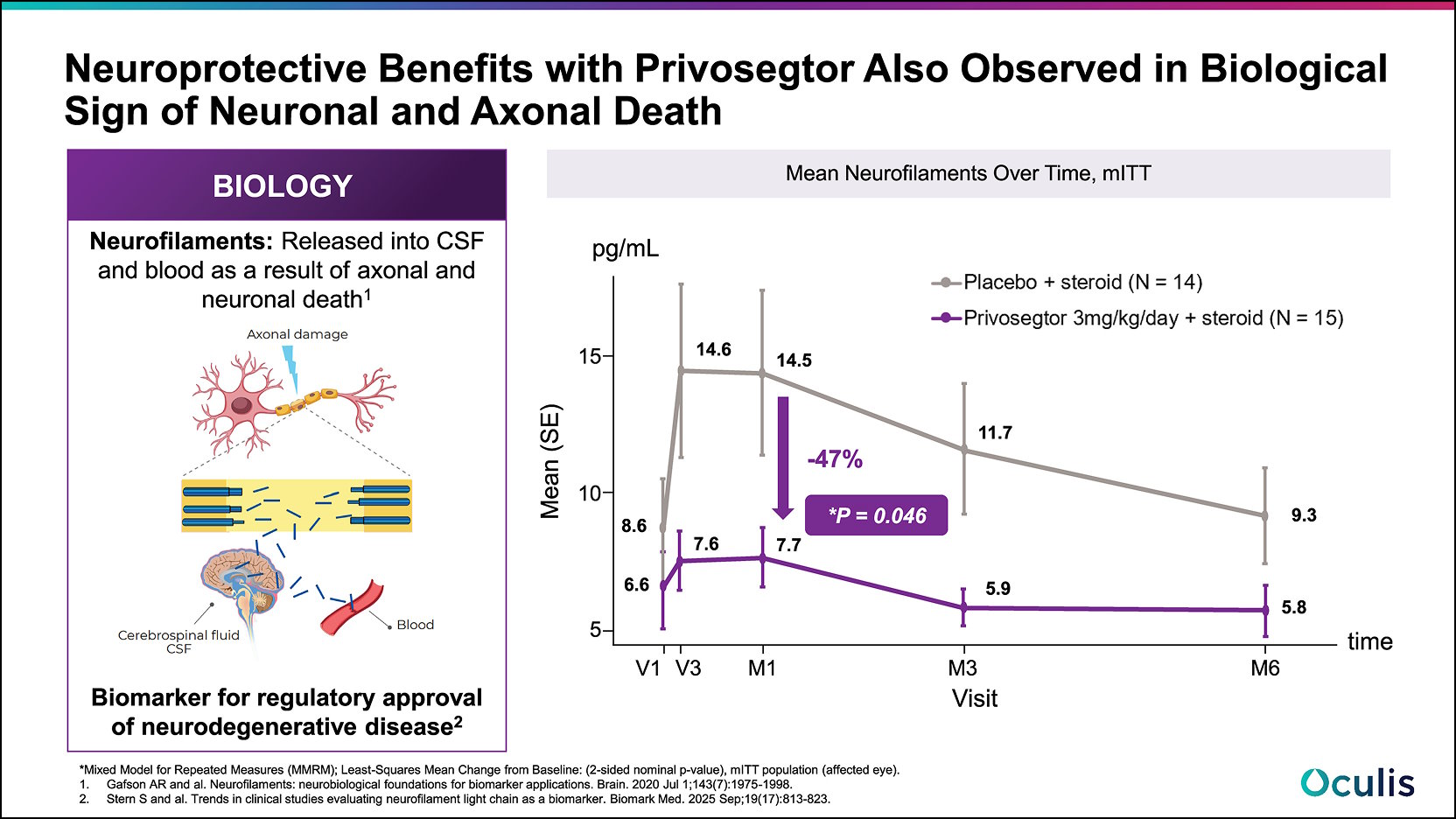
Secondary Efficacy Endpoint Assessed Biological Sign of Neuronal and Axonal Death:
Patients in the Privosegtor arm showed a positive effect on a biological sign of neuronal and axonal death with lower neurofilaments release in the bloodstream. Neurofilaments are released into the cerebrospinal fluid and blood as a result of axonal injury or neuronal death.
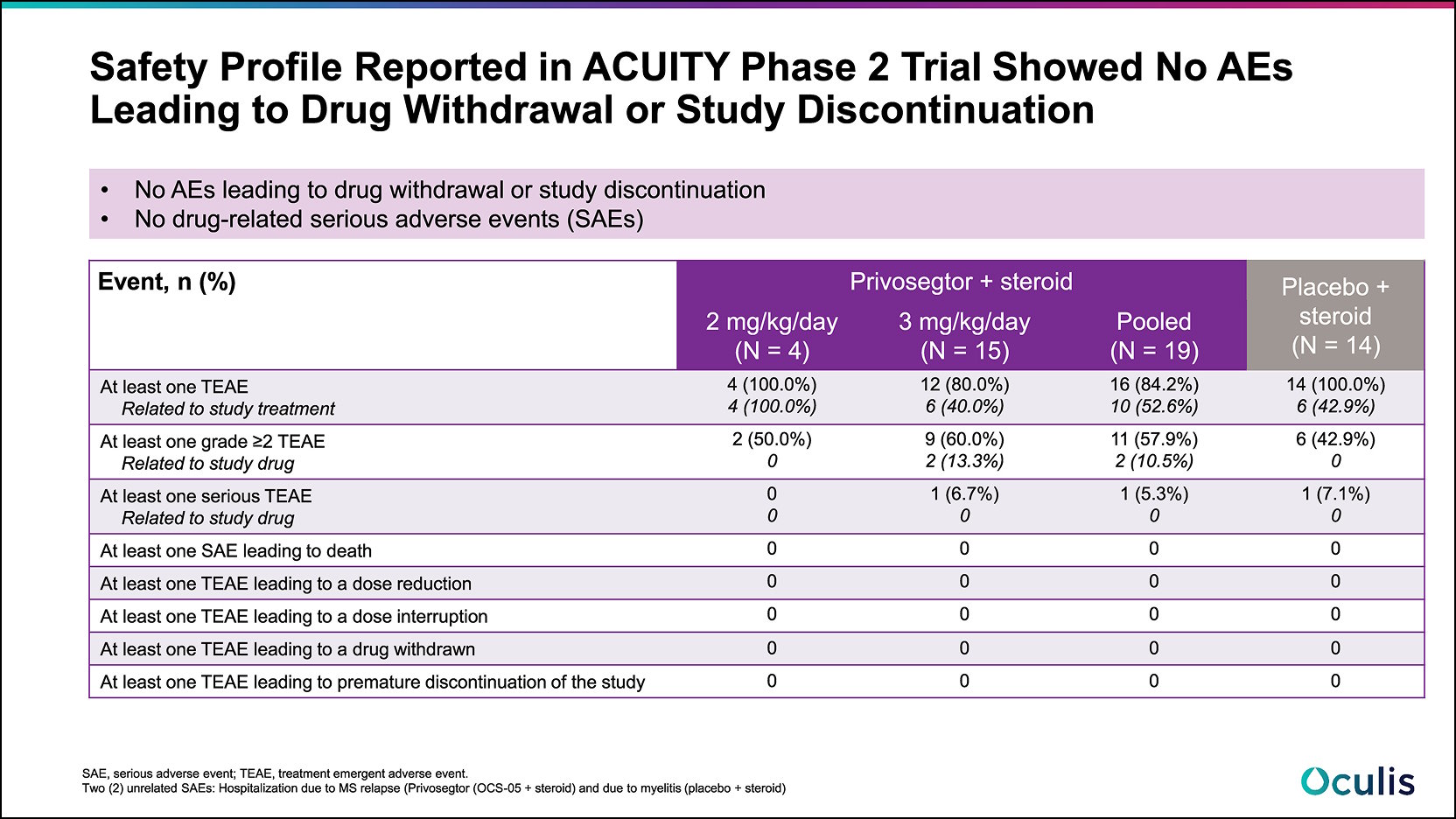
Treatment emergent adverse events (TEAEs):
- No drug-related serious adverse events (SAEs).
- No AEs leading to drug withdrawal or study discontinuation.
- Most frequently reported drug related AEs > 10% in the Privosegtor + steroid treatment group were headache: 2 patients (10.5%), and acne: 2 patients (10.5%).
Privosegtor has received Breakthrough Therapy designation from the U.S. Food and Drug Administration (FDA) and Orphan Drug designation from the U.S. FDA and the European Medicines Agency (EMA) for Optic Neuritis. To date there is no specific neuroprotective therapy approved for ON and unmet needs remain for therapies that can prevent vision loss after an acute episode of optic neuritis.
Oculis is not responsible for the content of any third-party sites or resources, and cannot guarantee the quality of information or support provided.
Privosegtor is an investigational drug candidate and has not been approved by the FDA. Safety and efficacy of Privosegtor for optic neuritis have not been established.
* Mixed Model for Repeated Measures (MMRM) Least-Squares Mean Change from Baseline: (nominal directional p- value), mITT population (study eye)
^ Mixed Model for Repeated Measures (MMRM) Least-Squares Mean Change from Baseline: (nominal p- value), mITT population (study eye)
